Finding valence electrons is crucial for understanding chemical bonding and reactivity. At HOW.EDU.VN, we provide expert insights into complex scientific concepts. Discover effective strategies to identify valence electrons and enhance your knowledge of atomic structure and chemical interactions. Uncover the secrets of electron configuration, the periodic table, and their applications in predicting chemical behavior.
1. Understanding Valence Electrons
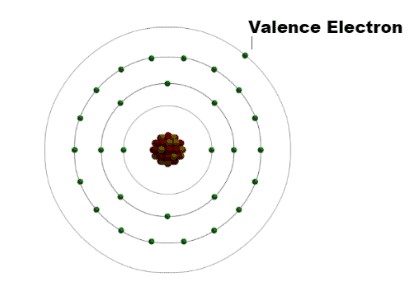
Before diving into methods for finding valence electrons, it’s essential to grasp what they are and why they matter. Valence electrons are the outermost electrons of an atom, residing in what’s known as the valence shell. These electrons are responsible for an atom’s chemical behavior, determining how it will interact with other atoms to form chemical bonds.
1.1. Defining Key Terms
To fully comprehend the concept of valence electrons, let’s define some essential terms:
-
Valence: The number of electrons an atom needs to gain or lose to achieve a stable electron configuration, resembling that of a noble gas.
-
Valence Electrons: The electrons located in the outermost shell of an atom, which dictate the atom’s stability and bonding behavior.
-
Valence Shell: The outermost energy level of an atom, which can be either occupied or unoccupied by electrons.
1.2. The Significance of Valence Electrons
Valence electrons play a pivotal role in determining an atom’s chemical properties and its ability to form bonds with other atoms. The number of valence electrons an atom possesses influences whether it will behave as a metal or a nonmetal and dictates the types of chemical bonds it can form. Understanding valence electrons is, therefore, fundamental to understanding chemical reactions and the formation of molecules.
1.3. The Role of Valence Electrons in Chemical Bonding
Valence electrons are the key players in chemical bonding. Atoms tend to gain, lose, or share valence electrons to achieve a stable electron configuration, typically with eight valence electrons (the octet rule) or two valence electrons (for elements like hydrogen and helium). This drive for stability leads to the formation of ionic, covalent, and metallic bonds, which hold molecules and compounds together.
2. Methods for Finding Valence Electrons
There are two primary methods for determining the number of valence electrons in an atom: using the periodic table and using electron configuration. Each method offers a unique approach, and understanding both will provide a comprehensive understanding.
2.1. Using the Periodic Table
The periodic table is a treasure trove of information about the elements, including the number of valence electrons. The periodic table method is a straightforward approach that relies on the organization of elements into groups (vertical columns) based on similar chemical properties.
2.1.1. How the Periodic Table Reveals Valence Electrons
The number of valence electrons for main group elements (Groups 1, 2, and 13-18) corresponds to the group number. For example, elements in Group 1 have one valence electron, elements in Group 2 have two valence electrons, and so on. This pattern arises because elements in the same group have similar electron configurations in their outermost shells, leading to similar chemical behavior.
2.1.2. Applying the Periodic Table Method
To find the number of valence electrons for an element using the periodic table, simply locate the element and identify its group number. The group number (for main group elements) directly corresponds to the number of valence electrons.
2.1.3. Examples of Using the Periodic Table to Find Valence Electrons
Let’s illustrate the periodic table method with a few examples:
-
Sodium (Na): Located in Group 1, sodium has one valence electron.
-
Magnesium (Mg): Located in Group 2, magnesium has two valence electrons.
-
Aluminum (Al): Located in Group 13, aluminum has three valence electrons.
-
Oxygen (O): Located in Group 16, oxygen has six valence electrons.
-
Chlorine (Cl): Located in Group 17, chlorine has seven valence electrons.
2.1.4. Limitations of the Periodic Table Method
The periodic table method is a quick and easy way to determine the number of valence electrons for main group elements. However, it does not apply to transition metals (Groups 3-12), which have more complex electron configurations and varying numbers of valence electrons. For transition metals, the electron configuration method is more reliable.
2.2. Using Electron Configuration
Electron configuration provides a detailed description of how electrons are arranged within an atom’s energy levels and orbitals. This method allows you to directly determine the number of valence electrons by examining the electron configuration of an element.
2.2.1. Understanding Electron Configuration
Electron configuration describes the specific arrangement of electrons within an atom’s energy levels and sublevels (orbitals). The electron configuration follows specific rules, such as the Aufbau principle (electrons fill the lowest energy levels first), Hund’s rule (electrons individually occupy orbitals within a sublevel before pairing up), and the Pauli exclusion principle (no two electrons can have the same set of four quantum numbers).
2.2.2. Writing Electron Configurations
To write the electron configuration of an atom, you need to know the number of electrons it possesses. This is equal to the atom’s atomic number, which can be found on the periodic table. Then, follow the filling order of orbitals, which is:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p
Each orbital can hold a maximum number of electrons: s orbitals hold 2 electrons, p orbitals hold 6 electrons, d orbitals hold 10 electrons, and f orbitals hold 14 electrons.
2.2.3. Identifying Valence Electrons from Electron Configuration
Once you have written the electron configuration of an atom, the number of valence electrons can be determined by identifying the electrons in the outermost energy level (the highest principal quantum number). Add up the number of electrons in the s and p orbitals of the outermost energy level to find the total number of valence electrons.
2.2.4. Examples of Using Electron Configuration to Find Valence Electrons
Let’s illustrate the electron configuration method with a few examples:
-
Sodium (Na): The electron configuration of sodium is 1s²2s²2p⁶3s¹. The outermost energy level is 3, and it contains one electron in the 3s orbital. Therefore, sodium has one valence electron.
-
Oxygen (O): The electron configuration of oxygen is 1s²2s²2p⁴. The outermost energy level is 2, and it contains two electrons in the 2s orbital and four electrons in the 2p orbital. Therefore, oxygen has six valence electrons (2 + 4 = 6).
-
Chlorine (Cl): The electron configuration of chlorine is 1s²2s²2p⁶3s²3p⁵. The outermost energy level is 3, and it contains two electrons in the 3s orbital and five electrons in the 3p orbital. Therefore, chlorine has seven valence electrons (2 + 5 = 7).
2.2.5. Advantages of the Electron Configuration Method
The electron configuration method is a more detailed and accurate way to determine the number of valence electrons, especially for transition metals and elements with complex electron configurations. It provides a complete picture of how electrons are arranged within an atom, allowing for a deeper understanding of its chemical behavior.
3. Valence Electrons in Molecules
Understanding valence electrons is not only important for individual atoms but also for molecules. The number of valence electrons in a molecule determines how it will interact with other molecules and its overall chemical properties.
3.1. Calculating Valence Electrons in Molecules
To calculate the total number of valence electrons in a molecule, simply add up the number of valence electrons for each atom in the molecule. This total number of valence electrons is crucial for drawing Lewis structures and predicting the molecule’s bonding and reactivity.
3.2. Examples of Calculating Valence Electrons in Molecules
Let’s illustrate the calculation of valence electrons in molecules with a few examples:
-
Water (H₂O): Each hydrogen atom has one valence electron, and the oxygen atom has six valence electrons. Therefore, the total number of valence electrons in a water molecule is (2 x 1) + 6 = 8.
-
Carbon Dioxide (CO₂): The carbon atom has four valence electrons, and each oxygen atom has six valence electrons. Therefore, the total number of valence electrons in a carbon dioxide molecule is 4 + (2 x 6) = 16.
-
Methane (CH₄): The carbon atom has four valence electrons, and each hydrogen atom has one valence electron. Therefore, the total number of valence electrons in a methane molecule is 4 + (4 x 1) = 8.
3.3. Using Valence Electrons to Draw Lewis Structures
Lewis structures are diagrams that show the bonding between atoms in a molecule, as well as any lone pairs of electrons. The number of valence electrons is essential for drawing accurate Lewis structures.
3.3.1. Steps for Drawing Lewis Structures
-
Calculate the total number of valence electrons in the molecule.
-
Draw the skeleton structure of the molecule, connecting atoms with single bonds.
-
Distribute the remaining valence electrons as lone pairs around the atoms, starting with the most electronegative atoms, to satisfy the octet rule (or duet rule for hydrogen).
-
If any atoms do not have an octet, form multiple bonds (double or triple bonds) to share electrons and satisfy the octet rule.
3.3.2. Example of Drawing a Lewis Structure
Let’s draw the Lewis structure for carbon dioxide (CO₂):
-
The total number of valence electrons is 16.
-
Draw the skeleton structure: O-C-O
-
Distribute the remaining valence electrons as lone pairs around the oxygen atoms: O=C=O with each oxygen having two lone pairs.
-
Each atom now has an octet, so the Lewis structure is complete.
4. Practical Applications of Valence Electrons
Understanding valence electrons has numerous practical applications in chemistry and related fields. From predicting chemical reactivity to designing new materials, valence electrons play a central role.
4.1. Predicting Chemical Reactivity
The number of valence electrons an atom possesses directly influences its chemical reactivity. Atoms with incomplete valence shells tend to be more reactive, as they seek to gain, lose, or share electrons to achieve a stable electron configuration.
4.2. Designing New Materials
Valence electrons are crucial in designing new materials with specific properties. By understanding how valence electrons interact, scientists can create materials with desired electrical conductivity, optical properties, and mechanical strength.
4.3. Understanding Chemical Reactions
Valence electrons are the key players in chemical reactions. Chemical reactions involve the rearrangement of valence electrons, leading to the formation of new chemical bonds and the creation of new molecules.
4.4. Developing New Technologies
The understanding of valence electrons is essential in developing new technologies, such as solar cells, batteries, and catalysts. These technologies rely on the manipulation of valence electrons to achieve specific functions.
5. Common Mistakes to Avoid
When working with valence electrons, it’s important to avoid common mistakes that can lead to incorrect results.
5.1. Confusing Valence Electrons with Core Electrons
Valence electrons are the electrons in the outermost shell of an atom, while core electrons are the electrons in the inner shells. It’s important to distinguish between these two types of electrons, as only valence electrons participate in chemical bonding.
5.2. Incorrectly Applying the Periodic Table Method
The periodic table method is a quick and easy way to determine the number of valence electrons for main group elements. However, it does not apply to transition metals. Make sure to use the electron configuration method for transition metals.
5.3. Making Errors in Electron Configuration
Writing the electron configuration of an atom requires following specific rules and filling the orbitals in the correct order. Making errors in electron configuration can lead to incorrect determination of valence electrons.
5.4. Ignoring the Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a stable electron configuration with eight valence electrons (or two for hydrogen and helium). Ignoring the octet rule can lead to incorrect Lewis structures and predictions of chemical behavior.
6. Real-World Examples
The concept of valence electrons is not just a theoretical concept but has real-world implications in various fields.
6.1. The Reactivity of Alkali Metals
Alkali metals (Group 1) have one valence electron, which they readily lose to form positive ions. This makes them highly reactive, as they easily react with nonmetals to form ionic compounds.
6.2. The Stability of Noble Gases
Noble gases (Group 18) have eight valence electrons (except for helium, which has two), making them very stable and unreactive. Their full valence shells make them resistant to forming chemical bonds.
6.3. The Bonding in Water Molecules
Water molecules (H₂O) are held together by covalent bonds formed by the sharing of valence electrons between oxygen and hydrogen atoms. The unique properties of water, such as its high boiling point and ability to act as a solvent, are due to the arrangement of valence electrons in the molecule.
6.4. The Conductivity of Metals
Metals are good conductors of electricity because their valence electrons are delocalized, meaning they are not bound to individual atoms but can move freely throughout the metal. This allows for the easy flow of electric current.
7. How HOW.EDU.VN Can Help You Master Chemistry
At HOW.EDU.VN, we understand that chemistry can be challenging. That’s why we offer expert guidance and resources to help you master complex concepts like valence electrons. Our team of experienced PhDs is dedicated to providing clear, concise, and accurate information to support your learning journey.
7.1. Access to Expert PhDs
Connect directly with leading PhDs specializing in chemistry and related fields. Get personalized answers to your specific questions and receive expert guidance tailored to your learning needs.
7.2. Customized Learning Solutions
Receive customized learning solutions designed to address your unique learning style and goals. Our experts can help you develop a personalized study plan, identify areas for improvement, and provide targeted support to help you succeed.
7.3. Simplified Complex Concepts
We break down complex concepts into easy-to-understand explanations and examples. Our goal is to make chemistry accessible to everyone, regardless of their background or experience.
7.4. 24/7 Availability
Access our resources and support anytime, anywhere. Whether you’re studying late at night or need help on the go, we’re here to assist you every step of the way.
8. Frequently Asked Questions (FAQs)
8.1. How to Calculate the Valence Electrons?
We can calculate the valence electrons by applying one of the following methods:
-
Periodic Table: Using the periodic table, we can determine the number of valence electrons by following these rules:
- All elements in a single vertical column have the same number of valence electrons.
- The atom’s main group number (group number excluding transition metals) indicates the number of valence electrons for these atoms.
For example, elements in Group 1 have 1 valence electron, those in Group 2 have 2 valence electrons, and so on.
We can find the valence electrons by applying the previous rules and observing the number of the group on the periodic table that contains the desired element.
-
Electron Configuration: Electron configuration refers to the distribution of electron in the orbitals (atomic or molecular).
After determining the electron configuration you can find the valence electrons by examining the number of electrons in the outermost shell (valence shell) which is equal to the number of valency.
8.2. What is the 2 8 8 18 Rule in Chemistry?
The 2 8 8 18 rule in chemistry refers to the electron filling rule or the electron configuration in the shells of an atom.
-
Electron Configuration: Electronic configuration is an arrangement of electrons in various shells, subshells and orbitals in an atom. It is written as the following pattern: 2, 8, 8, 18, 18, 32
-
Notation: It is written as nl x where:
- n indicates the principal quantum number
- I indicates the azimuthal quantum number or sub-shell
- x is the number of electrons.
-
Number of electrons : The number of electrons in n shell is given by the following equation:
2n²
e.g. in the first shell the number of electrons = 2.
8.3. Where to Find the Number of Valence Electrons on the Periodic Table?
We can find the number of valence electrons by observing the number of the group (vertical column) on the periodic table that contains the wanted element.
For more clarification, it is known that:
- Group 1 — 1 valence electron
- Group 2 — 2 valence electrons
- Group 13 — 3 valence electrons
- Group 14 — 4 valence electrons
- Group 15— 5 valence electrons
- Group 16— 6 valence electrons
- Group 17— 7 valence electrons
- Group 18 — 8 valence electrons (except for helium, which has 2)
For example:
The valence electrons of carbon (C) is 4 because on the periodic table, carbon is located in group 14 and by applying the role “The atom’s main group number is equal to the number of valence electrons for these atoms”. So, the valence electrons of carbon is 4.
The valence electrons of lithium (Li) is 1 because on the periodic table, Lithium is located in group 1 and by applying the role “The atom’s main group number is equal to the number of valence electrons for these atoms”. So, the valence electrons of Lithium is 1.
If we check iodine (I) in the periodic table, we will find it in group 17 and by applying the fact “The atom’s main group number is equal to the number of valence electrons for these atoms”.
So, the number of Iodine valence electrons is 7.
Remember that the periodic table method for finding the valence electrons can’t be used with the transition metals which are located in groups no( 3,4,5,6,7,8,9,10,11,12) because the atomic structure of these elements is different and needs a special method to calculate their valence electrons.
8.4. How Do You Determine the Number of Valence Electrons for Beryllium?
We can determine the number of valence electrons for Beryllium by applying the periodic table method:-
If we check beryllium (Be) in the periodic table, we will find it in group 2 and by applying the role “The atom’s main group number is equal to the number of valence electrons for these atoms”.
So, the number of valence electrons for beryllium is 2.
8.5. Can Valence Electrons Determine an Element’s Reactivity?
Yes, valence electrons are the primary factor in determining an element’s reactivity. Elements with a full valence shell, like noble gases, are generally unreactive because they do not need to gain, lose, or share electrons to achieve stability. Elements with incomplete valence shells are more reactive as they seek to achieve a stable electron configuration.
8.6. How Do Valence Electrons Contribute to Chemical Bonds?
Valence electrons are directly involved in forming chemical bonds between atoms. Atoms may transfer valence electrons to form ionic bonds or share valence electrons to form covalent bonds. The type of bond formed depends on the electronegativity difference between the atoms involved.
8.7. What Role Do Valence Electrons Play in Metallic Bonding?
In metallic bonding, valence electrons are delocalized, meaning they are not associated with individual atoms but are free to move throughout the metal lattice. This “sea” of electrons is responsible for the unique properties of metals, such as their high electrical and thermal conductivity.
8.8. Are Valence Electrons the Same as Free Electrons?
The terms “valence electrons” and “free electrons” are sometimes used interchangeably, but they have slightly different meanings. Valence electrons are the electrons in the outermost shell of an atom, while free electrons are valence electrons that are not bound to individual atoms and are free to move throughout a material. In metals, valence electrons are also free electrons, which contribute to electrical conductivity.
8.9. How Do You Determine the Number of Valence Electrons in an Ion?
To determine the number of valence electrons in an ion, start with the number of valence electrons in the neutral atom. If the ion is positively charged (cation), subtract the number of electrons lost from the number of valence electrons in the neutral atom. If the ion is negatively charged (anion), add the number of electrons gained to the number of valence electrons in the neutral atom.
8.10. How Can I Predict the Number of Bonds an Atom Will Form Based on Its Valence Electrons?
The number of bonds an atom will typically form can be predicted based on its valence electrons and the octet rule. Atoms tend to form bonds to achieve a full valence shell with eight electrons (or two for hydrogen). For example, carbon has four valence electrons and typically forms four bonds to achieve an octet. Oxygen has six valence electrons and typically forms two bonds to achieve an octet.
9. Call to Action
Do you have questions about valence electrons or other chemistry topics? Are you struggling to grasp complex scientific concepts? Don’t hesitate to seek expert guidance from HOW.EDU.VN. Our team of experienced PhDs is here to provide personalized support and help you succeed.
Contact us today to schedule a consultation and unlock your full potential.
Address: 456 Expertise Plaza, Consult City, CA 90210, United States
WhatsApp: +1 (310) 555-1212
Website: HOW.EDU.VN
Let how.edu.vn be your trusted partner in mastering chemistry and achieving your academic goals. We are committed to providing you with the knowledge, resources, and support you need to excel.